Doctors and Hospitals Sued for Allegedly Unnecessary Cardiac Procedures; Claims Include Medical Malpractice and Medicare Fraud
 How These Cases Differ From Medical Malpractice
How These Cases Differ From Medical Malpractice
A series of lawsuits around the country have sought to involve the federal government in claims that doctors and hospitals performed unnecessary surgical procedures on patients, putting those patients’ lives at risk and, in some cases, allegedly causing patient deaths. These lawsuits, filed under the federal False Claims Act (FCA), differ from medical malpractice cases in that they are not based on any breach of a standard of medical care, but rather on whether performing an unnecessary procedure constitutes fraud against the government. While the cases do not directly result in recovery of damages for the affected patients or their families, they often serve to expose negligent practices among doctors and hospitals, providing an opportunity to gather evidence for a medical malpractice claim.
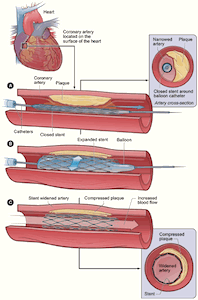
The U.S. Justice Department has settled claims with eleven hospitals in recent years alleging overuse of stents, small mesh tubes implanted into cardiac patients’ blood vessels to relieve blockage in a procedure known as angioplasty. Many hospitals maintain catheterization labs, or “cath labs,” to perform these procedures. A recent report from the American Medical Association and the Joint Commission described ten percent of elective angioplasties as “inappropriate” and one-third as “questionable.” Stents reportedly provide a substantial income stream for hospitals, leading to allegations that unnecessary procedures are motivated by profit. When severe complications result, such as torn arteries, internal bleeding, or blood clots, it highlights a conflict between the business motives of for-profit hospitals and the standard of care for medical professionals.
Different Ways The Legal System Holds Hospitals and Doctors Accountable
Patients or their families generally assert legal claims for medical malpractice, which might include injuries caused by an unnecessary stent. In cases involving the FCA, the initial complainants are usually insiders, such as a doctor or other hospital worker, who act as “whistleblowers” to expose fraudulent activity. A whistleblower files a qui tam lawsuit on behalf of the United States, claiming fraud against a government program like Medicare or Medicaid. The federal government may directly intervene in a qui tam lawsuit, or the qui tam plaintiff may continue to pursue it individually.
A qui tam lawsuit requires proof that a doctor or other medical professional submitted false claims for procedures, such as, by demonstrating that the procedures were not medically necessary. This differs from the proof required in a medical malpractice claim, that a doctor breached a duty of care, causing injury to a patient. Still, a whistleblower suit can be helpful in building a case for medical malpractice.
Examples of Whistleblower Cases
In a Georgia case filed in 2010, a hospital cath lab nurse sued the hospital and a doctor who worked in the cath lab. The nurse alleged the doctor was not qualified to perform catheterization procedures, and that his lack of qualifications injured multiple patients and possibly caused the death of one patient. The government intervened in the lawsuit, and it settled with the hospital for $840,000 in 2012. A cardiologist in Pennsylvania filed suit against five colleagues, accusing them of billing Medicare for unnecessary angioplasty procedures resulting in at least one death. The government declined to intervene in that case.
Contact An Experienced Medical Malpractice Law Firm Today!
Wais, Vogelstein, Forman, Koch & Norman’s team of medical malpractice attorneys has represented clients in Maryland for over twenty years, helping them recover damages for injuries resulting from the negligence of doctors, pharmacists, and other medical professionals. We have recovered millions of dollars in damages for our clients through verdicts and settlements. We are available 24/7 and can visit you in your home or at the hospital.
To schedule a free and confidential consultation, contact us today online, at (410) 567-0800.
